Regeneration of olfactory neuroepithelium in 3-methylindole-induced anosmic rats treated with intranasal chitosan (Li et al., 2021)1
Anosmia has received a lot of attention recently, as a common side effect of Covid. And it’s a scary prospect! What I hadn’t realised is just how common it is even under normal circumstances – around 3.2% of US adults are anosmic, rising to ~20% of over 60s.2 It’s also a tough condition to detect: it’s not included in standard physical exams, and so won’t be detected until a patient self-reports specific symptoms. Given how difficult it is to notice a gradual decline in sensitivity to smells, that’s only likely when it becomes severe.
The most common causes of anosmia are trauma and post-infection “hangovers”, both of which come on suddenly, gradually onsetting sinonasal disorders, or congenital defects. See figure 1 for a quick diagnostic flow chart.
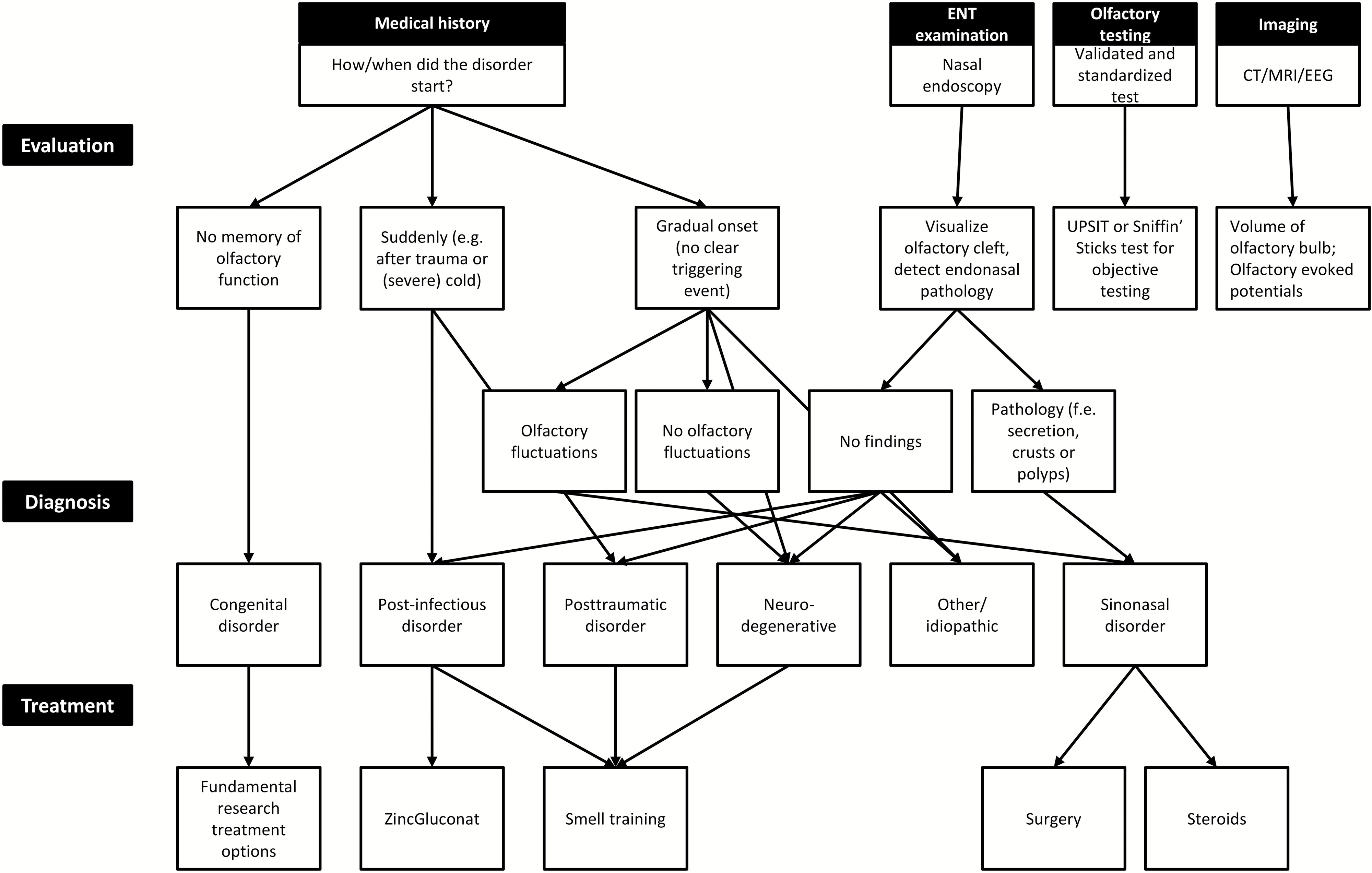 |
|---|
| Figure 1: Diagnosis and Treatments, reproduced from Boesvelt et al. (2017)2 |
In almost every case the treatment options aren’t great: for example, ZincGluconat in combination with steroids raises recovery rates to just 28% for anosmia following traumatic head injury3, and smell training (with “Sniffin’ Sticks”) improves recovery in ~30% of post-infection cases.4 So, new treatments are certainly needed!
Olfactory Epithelium
The olfactory epithelium is a pretty fascinating patch of epithelial tissue, covering about 4.5cm2 per nostril, deep into the nasal cavity (it’s about 7cm behind your nostril – so well inside your head, rather than your nose!).5 Olfactory neurons are integrated into the epithelium, and turn over every 30-60 days2 (sniffing is a dangerous job for a cell, I guess). Those neurons are continually replaced by the multipotent neural progenitor Globose basal cells.6 Globose basal cells are transit amplifying cells, generated by the slowly dividing Horizontal basal cells, which exist in an ECM niche. Horizontal basal cells can produce all the cell types in the olfactory epithelium, giving them significant regenerative potential.
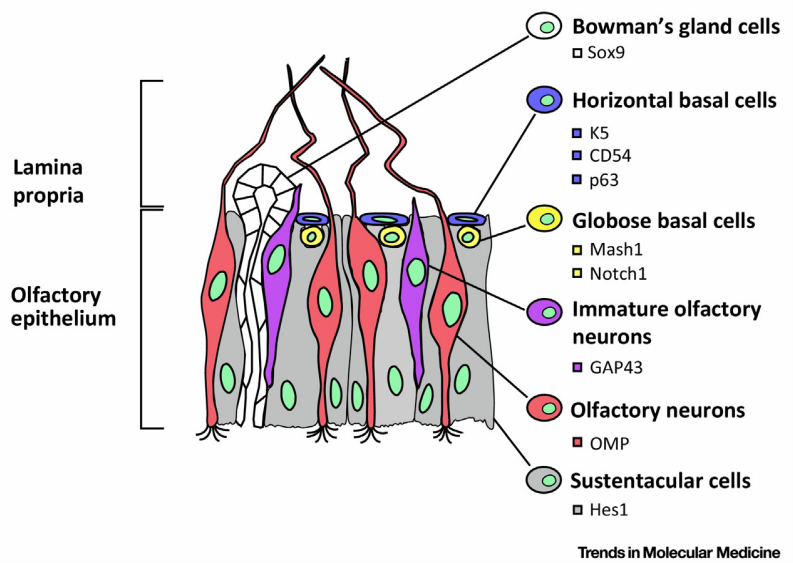 |
|---|
| Figure 2: Olfactory Neural Epithelium, reproduced from Lavoie et al., 20176 |
What’s this paper trying to do then?
So, on to the work at hand. Previous work by the authors has shown that the polysaccharide chitosan promotes the differentiation of neural epithelial cells from rat embryos into neurons.7 In this paper, Li et al. evaluate the ability of chitosan solutions to regenerate tissue and improve sensory performance in live animals.
How else might they go about it?
Surprisingly, to me, there doesn’t seem to be that much work on neural epithelial regeneration. Neural epithelial tissue, including stem cells, can be obtained readily by biopsy, or more recently with a brush swab gentle enough not to require local anaesthetic, and an animal model is available (as here) by injecting rats or mice with 3-methylindole (skatole) to induce chemical lesion of the neural epithelium.6,8 I would expect this accessibility, combined with the interesting regenerative abilities of the tissue, to attract more interest than the small number of studies looking at promoting regeneration with small molecule drugs (e.g. statins9, lipoic acid10) or Ginkgo Biloba.11 One interesting strategy which doesn’t seem to have been explored recently is the activation of horizontal basal cells, which become quiescent with age, into the rapidly dividing globular basal cells,2 which has been shown to be promoted by Pax6.12
What did they find?
Rats treated twice a week with intranasal chitosan (see Hanson et al., 2013, for a pretty amazing video on how to get rodents to put up with this treatment13) recovered a functional sense of smell, measured by performance in a food maze, over 28 days. A control group treated with buffer solution showed no real recovery of performance over the same period.
The treated animals showed both better bulk tissue recovery (from an epithelial layer 14µm after methylindole treatment to 60µm after 28 days, the same as a normal mouse, against recovery to just 27 µm over the same period without chitosan) and maturation of mature olfactory receptor neurons (ORN). Mature ORNs are identified by fluorescent staining of the protein OMP (olfactory marker protein), confirmed by Western blotting to prove increased OMP expression.
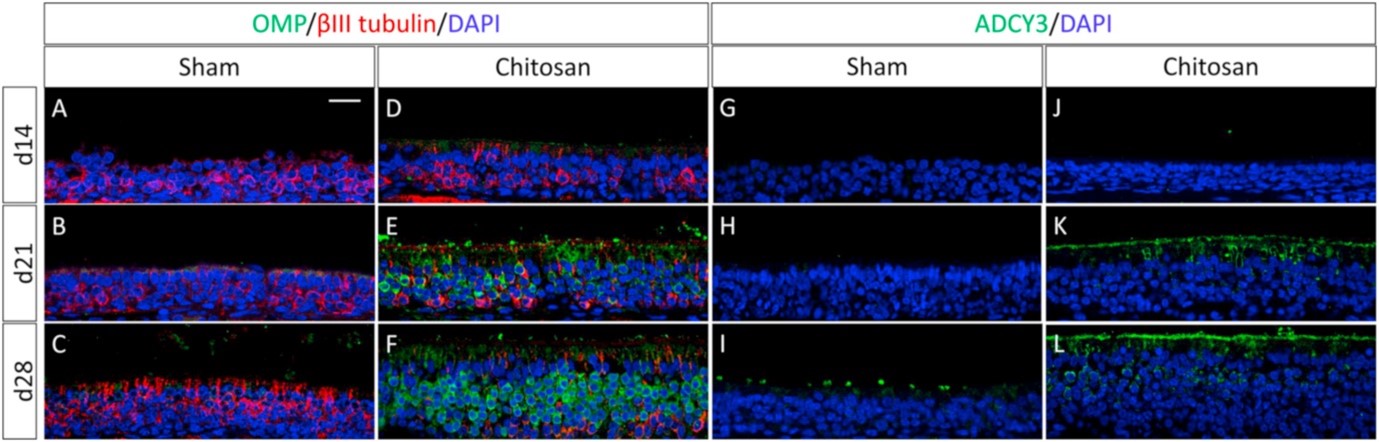 |
|---|
| Figure 3: Presence of Mature ORNs in chitosan-treated rats, reproduced from Li et al., 20211 |
What’s next?
I was quite taken with this work. Chitosan is readily available as a food supplement, suggesting it’s really quite safe, and intranasal delivery is obviously a good starting point for an anosmia treatment. The real question is, can we use it to make an ordinary person (me!) into a supertaster?
References
- Li, S.-T., Young, T.-H. & Huang, T.-W. Regeneration of olfactory neuroepithelium in 3-methylindole-induced anosmic rats treated with intranasal chitosan. Biomaterials 120738 (2021) doi:10.1016/j.biomaterials.2021.120738.
- Boesveldt, S. et al. Anosmia—A Clinical Review. Chemical Senses 42, 513–523 (2017).
- Jiang, R.-S., Twu, C.-W. & Liang, K.-L. Medical treatment of traumatic anosmia. Otolaryngol Head Neck Surg 152, 954–958 (2015).
- Damm, M. et al. Olfactory training is helpful in postinfectious olfactory loss: a randomized, controlled, multicenter study. Laryngoscope 124, 826–831 (2014).
- Olfactory epithelium. Wikipedia (2020).
- Lavoie, J. et al. The Olfactory Neural Epithelium As a Tool in Neuroscience. Trends in Molecular Medicine 23, 100–103 (2017).
- Li, S.-T., Young, T.-H., Lin, C.-F. & Huang, T.-W. Promotion of olfactory receptor neuron differentiation of olfactory neuroepithelial cells by using chitosan solution. Am J Rhinol Allergy 31, 289–292 (2017).
- Peele, D. B. et al. Functional deficits produced by 3-methylindole-induced olfactory mucosal damage revealed by a simple olfactory learning task. Toxicology and Applied Pharmacology 107, 191–202 (1991).
- Kim, H. Y. et al. Effects of Statins on the Recovery of Olfactory Function in a 3-Methylindole–Induced Anosmia Mouse Model. Am J Rhinol�Allergy 26, e81–e84 (2012).
- Hummel, T., Heilmann, S. & Hüttenbriuk, K.-B. Lipoic Acid in the Treatment of Smell Dysfunction Following Viral Infection of the Upper Respiratory Tract. The Laryngoscope 112, 2076–2080 (2002).
- Lee, C. H., Mo, J.-H., Shim, S. H., Ahn, J.-M. & Kim, J.-W. Effect of Ginkgo Biloba and Dexamethasone in the Treatment of 3-methylindole-induced Anosmia Mouse Model. American Journal of Rhinology 22, 292–296 (2008).
- Packard, A. I., Lin, B. & Schwob, J. E. Sox2 and Pax6 Play Counteracting Roles in Regulating Neurogenesis within the Murine Olfactory Epithelium. PLoS One 11, e0155167 (2016).
- Hanson, L. R., Fine, J. M., Svitak, A. L. & Faltesek, K. A. Intranasal Administration of CNS Therapeutics to Awake Mice. J Vis Exp (2013) doi:10.3791/4440.